
For the science novice, the chemistry of cleaning can be tough to get your head around.
I never cared much for science in high school, so learning all about chemistry’s role in making your carpets clean, as part of my role as a blogger for E-Dry, has definitely been eye-opening.
The most important part about chemistry that’s relevant to effective cleaning is the pH Scale. Here’s what you need to know.
So, what is the pH Scale?
I’m sure you’ve heard of it before, but the scale is a bigger part of your life than you think.
pH stands for ‘potential of hydrogen’ and measures how many hydrogen ions are in a substance. Where that substance sits on the scale determines whether it is acidic, neutral or alkaline. Everything, from the make-up you put on your face this morning to the milk you have on your cereal, sits on the scale somewhere.
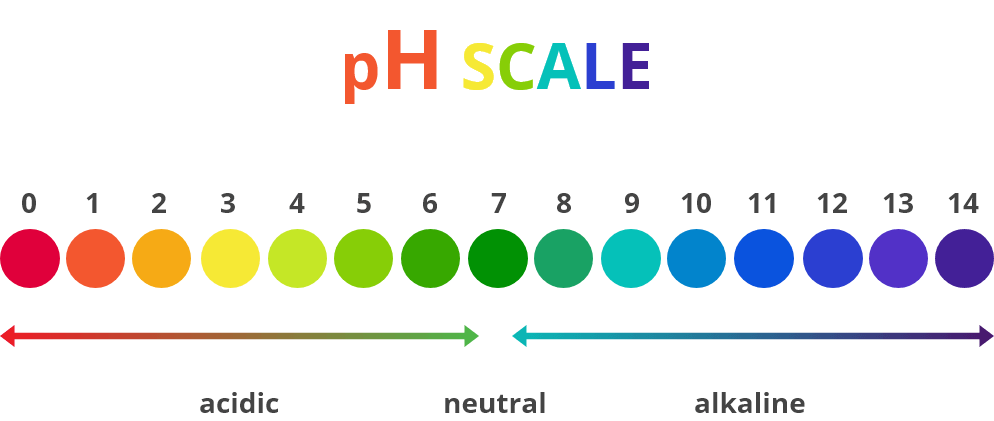
The scale has 14 points – a substance with a low concentration of hydrogen ions is acidic (0-6) and a high concentration is alkaline (8-14). A 7 on the scale is a neutral substance.
How You Use the Scale in Your Life

Any water-based substance has a pH rating, including food, drinks, beauty items and our favourite, cleaning products.
Here’s some everyday items below, and where they sit on the scale.
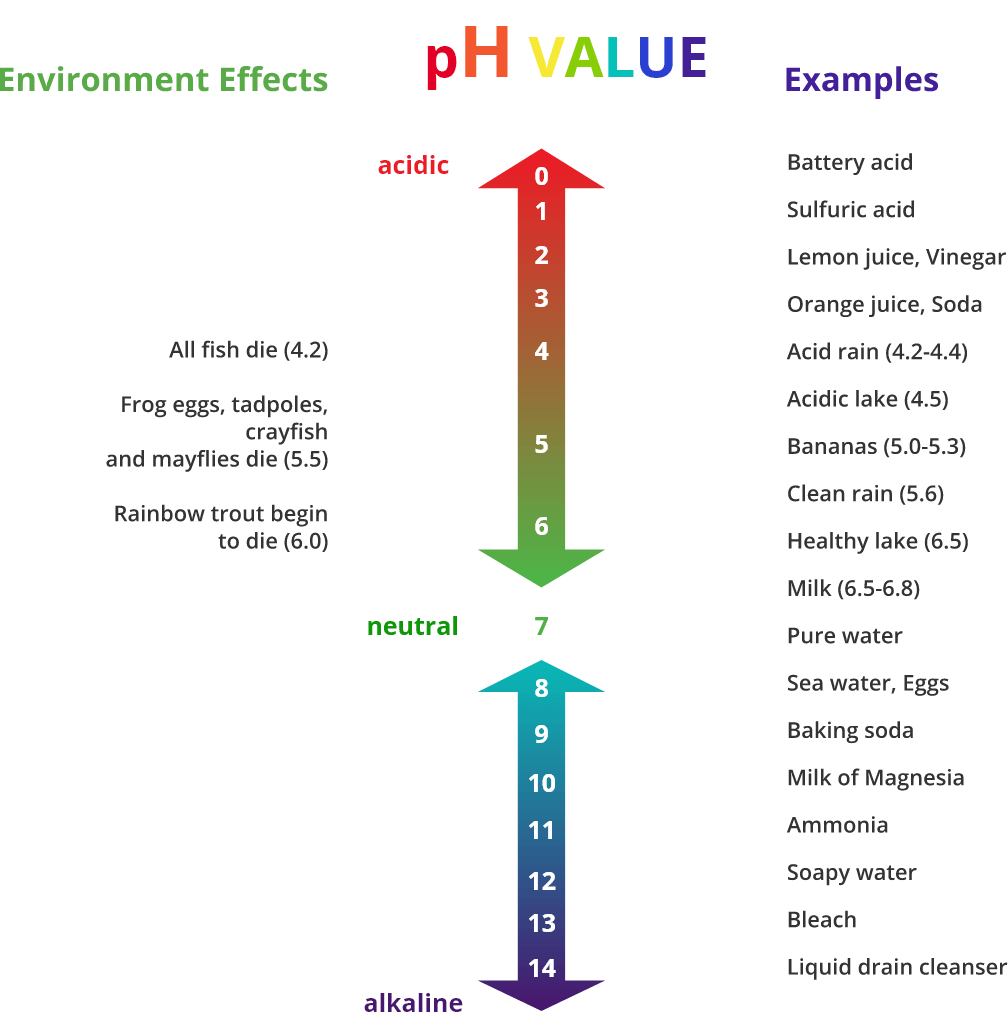
Source: saddlespace
Why You Should Consider the Scale When Cleaning
Many believe that strongly alkaline cleaners make the best cleaners but this isn’t necessarily the case.
When cleaning, you should aim to neutralise the object or the stain’s pH level, so the alkalinity or acidity of the soiled area you are cleaning. In simple terms, acid neutralises alkaline and vice versa.
Alkaline Cleaners
Contain potent bases including potassium hydroxide and sodium hydroxide.
Dissolves acidic substances such as fats, oils and grease (such as foods).
Known alkaline cleaners include bleach, oven cleaners and multi-purpose sprays.
Acidic Cleaners
Dissolves alkaline substances such as mineral deposits or rust, so they’re usually great for cleaning bathroom areas like toilets.
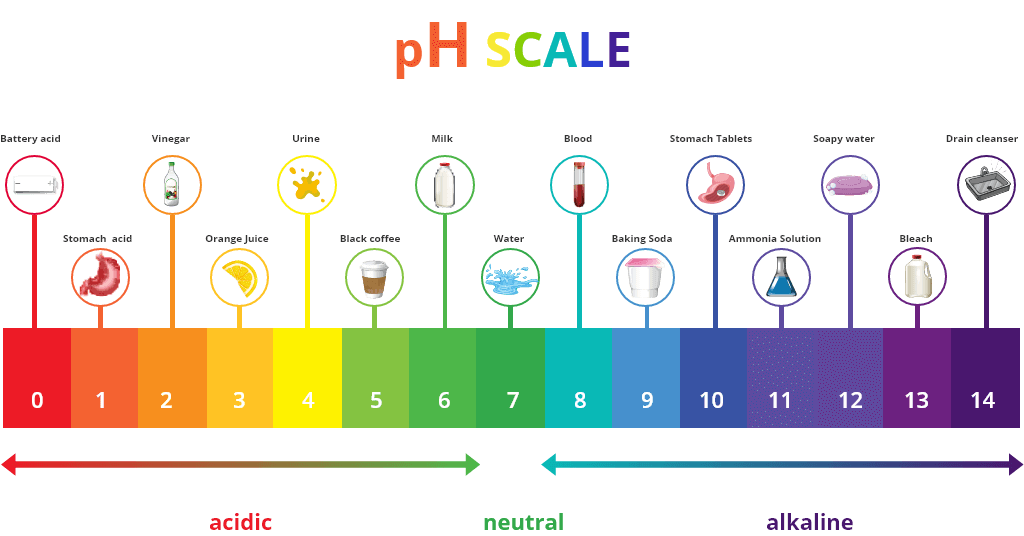
This diagram demonstrates why vinegar and bi-carb soda are among my favourite natural cleaning products. While they aren’t as toxic as other cleaners, they can still pack a punch.
|
Note: It is worth noting that highly alkaline cleaning products like drain cleaner or acidic like battery acid are reactive, which means that they can cause burns. |
How does E-Dry use the pH scale?
We use the pH scale to restore the chemical balance of your carpets, which is why it is so important for the technician to know what any carpet stains are and what you’ve already used to try to clean.
The E-Dry technician has experienced understanding of the pH balance and may just know how to get rid of that stain you thought would be impossible to remove.
What Your Professional Carpet Cleaner Can Teach You about Ph levels

You need a close to neutral substance to clean wool or stain-resistant fibres.
Extremely high or low Ph products can damage surfaces. For example, vinegar can etch marble and strong alkaline cleaners can irreversibly damage the dyes in your carpet).
The solvents used during carpet dry cleaning dissolves soil and is absorbed into the cleaning.
Dry cleaning uses less water and chemicals, so that no chemical residues are left behind and your carpets are walk-on-dry in just one hour.
SUBSCRIBE TO OUR NEWSLETTER
Sign up for email promotions, tips and special offers.



